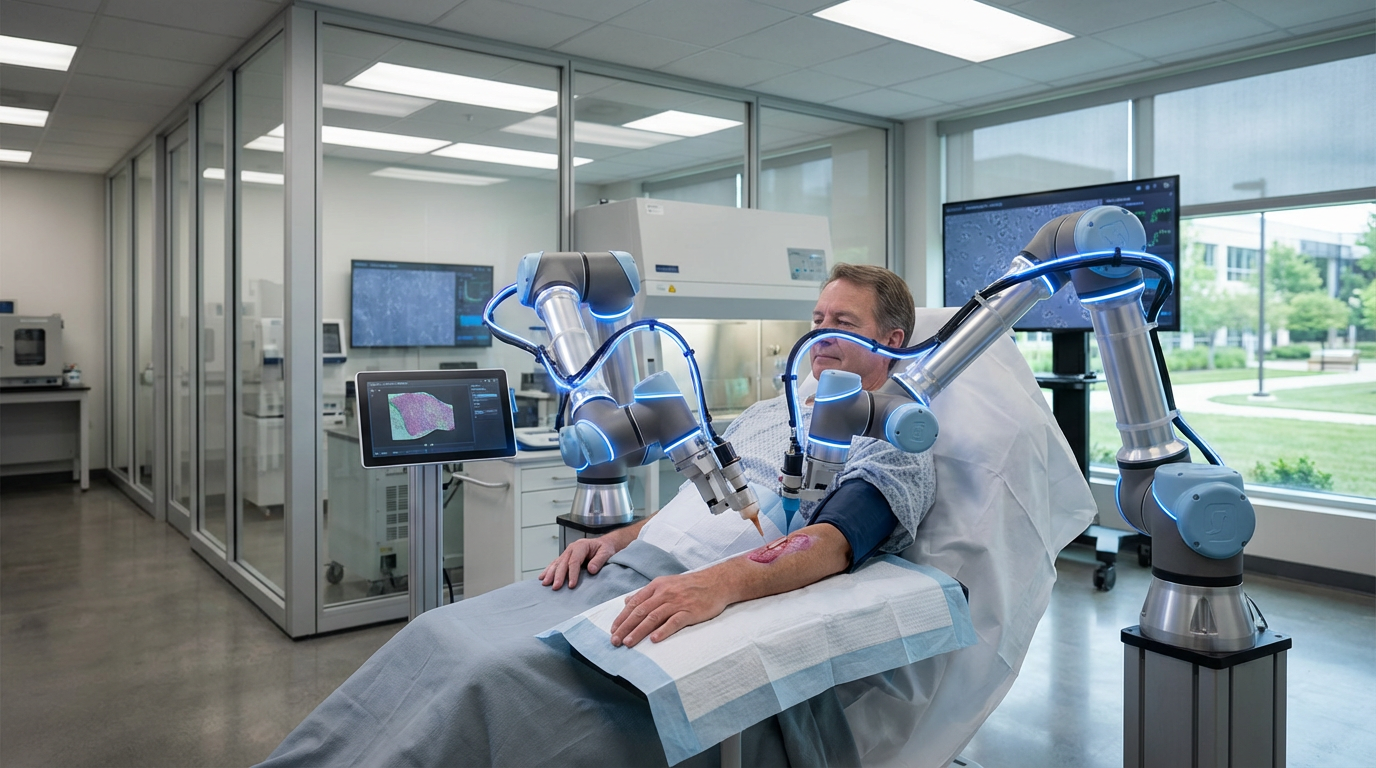
On-Demand Organoid Transplantation
On-demand organoid transplantation represents a paradigm shift in regenerative medicine, moving away from the traditional dependence on donor organs toward patient-specific biological solutions. The technology begins with the extraction of somatic cells—typically skin or blood cells—from the patient, which are then reprogrammed into induced pluripotent stem cells (iPSCs) through the introduction of specific transcription factors. These iPSCs possess the remarkable ability to differentiate into virtually any cell type in the human body. Through carefully controlled culture conditions, growth factors, and three-dimensional scaffolding systems, researchers guide these stem cells to self-organize into miniature, simplified versions of organs called organoids. These structures, ranging from a few millimeters to several centimeters in size, recapitulate key architectural features and cellular compositions of their full-sized counterparts, including liver lobules, kidney nephrons, or cortical brain tissue. The organoids develop functional capabilities such as metabolic processing, filtration, or neural signaling, though at a reduced scale compared to complete organs.
The primary challenge this technology addresses is the severe shortage of transplantable organs and the complications associated with immune rejection in traditional transplantation. Globally, hundreds of thousands of patients languish on waiting lists for organs that may never arrive, while those who do receive transplants face lifelong immunosuppression regimens that carry significant health risks. On-demand organoid transplantation circumvents these issues by using the patient's own genetic material, virtually eliminating rejection concerns. Rather than replacing entire organs, this approach enables targeted regeneration of specific damaged regions within failing tissues. For instance, liver organoids can be introduced into cirrhotic tissue to restore metabolic function in localized areas, while kidney organoids might supplement filtration capacity in patients with chronic renal disease. This strategy also opens new therapeutic avenues for conditions previously considered untreatable, such as neurodegenerative diseases where brain organoids could potentially replace lost neural circuits or support existing damaged networks.
Early clinical investigations and preclinical studies suggest promising outcomes, with research institutions exploring organoid transplantation for liver disease, kidney failure, and certain neurological conditions. Current deployments remain largely experimental, with regulatory frameworks still evolving to accommodate these novel biological therapies. The technology faces technical hurdles including vascularization—ensuring transplanted organoids receive adequate blood supply—and achieving full functional integration with host tissues. However, advances in bioprinting, microfluidic culture systems, and vascular engineering are progressively addressing these limitations. As the field matures, on-demand organoid transplantation may fundamentally transform the treatment landscape for organ failure, shifting the paradigm from scarce donor-dependent transplantation toward abundant, personalized regenerative solutions that extend both lifespan and healthspan by maintaining organ function throughout the aging process.
Related Organizations
A pioneering research institute where the technology to grow mini-organs (organoids) from adult stem cells was originally developed by the Clevers Lab.
A premier research institute led by Dr. Anthony Atala, famous for printing bladders and developing the ITOP printer.
Biotechnology company using lymph nodes as bioreactors to regrow functioning ectopic organs, including the thymus.
Home to the Center for Stem Cell and Organoid Medicine (CuSTOM), focusing on developing organoid-based therapies.
Developing stem cell-derived islet cell therapies (VX-880) for Type 1 Diabetes.
Develops the Cell Pouch System, an implantable medical device that creates a natural environment for therapeutic cells.
Develops C-Stem technology for mass-producing high-quality induced pluripotent stem cells (iPSCs) and micro-tissues.
Uses direct cellular conversion technology to transform cell types without passing through a pluripotent stem cell state.
Researchers here successfully transplanted human intestinal organoids into patients with inflammatory bowel disease.
A major pharmaceutical company actively investing in and establishing centers for organ-on-a-chip technology to replace animal testing.