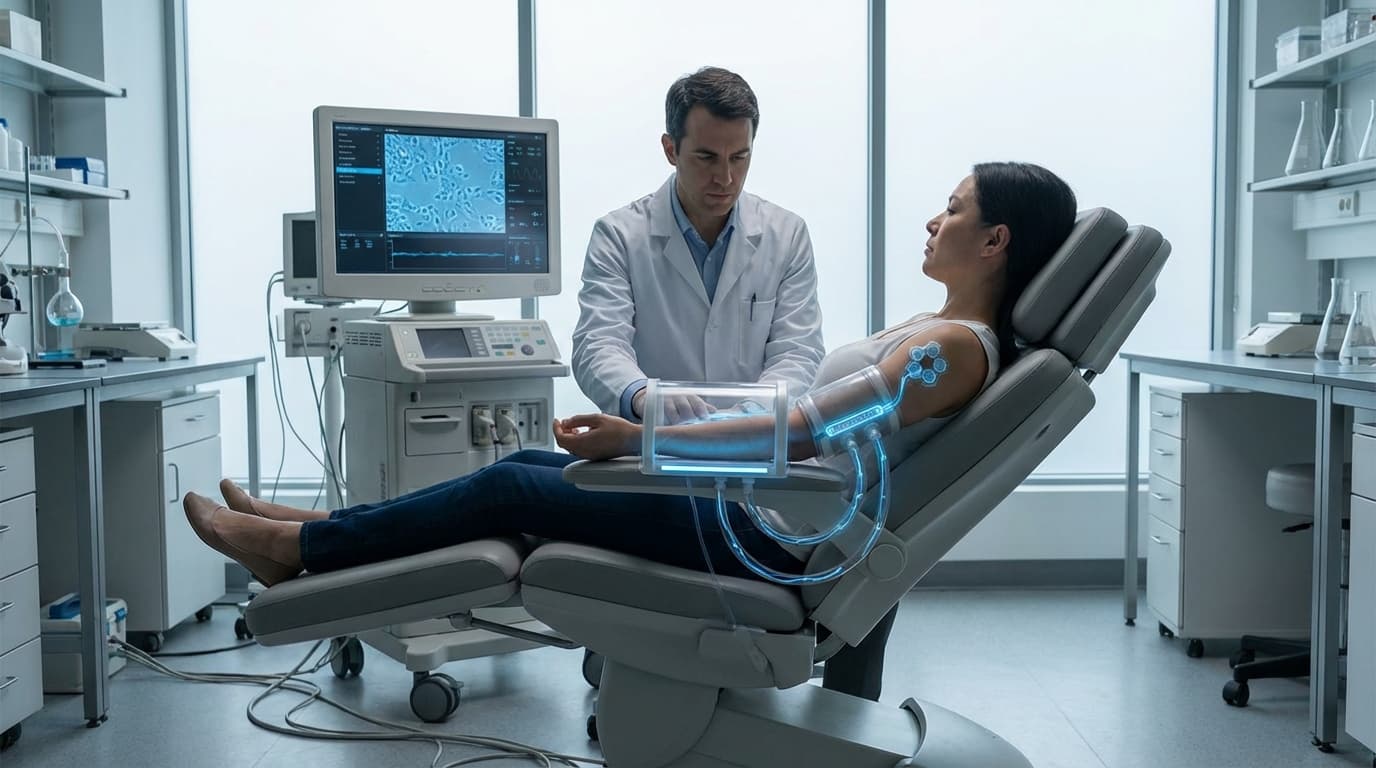
Mitochondrial Augmentation Microdevices
Mitochondrial dysfunction stands as one of the fundamental hallmarks of aging, contributing to cellular energy depletion, increased oxidative damage, and progressive tissue deterioration across multiple organ systems. As cells age, their mitochondria—the powerhouses responsible for generating cellular energy—accumulate mutations, lose efficiency, and produce harmful reactive oxygen species that accelerate further damage. Traditional therapeutic approaches have struggled to address this decline at its source, as delivering functional replacement mitochondria to aging tissues has proven technically challenging. Mitochondrial augmentation microdevices represent a novel bioengineering solution to this problem, offering a direct method to replenish cellular energy infrastructure. These miniaturized platforms integrate microfluidic channels, filtration systems, and precision injection mechanisms into implantable or catheter-delivered formats. The devices work by first harvesting mitochondria from healthy autologous tissue sources or from specially prepared donor cells, then enriching and concentrating these organelles while maintaining their functional integrity. Advanced membrane filtration and gentle centrifugation techniques separate viable mitochondria from cellular debris, creating purified preparations ready for therapeutic delivery. The injection component employs micro-needle arrays or targeted catheter systems to introduce these healthy mitochondria directly into aging tissues, where they can be taken up by recipient cells through natural endocytotic pathways or facilitated transfer mechanisms.
Research in regenerative medicine suggests that replacing dysfunctional mitochondria could address multiple age-related pathologies simultaneously, as mitochondrial decline contributes to conditions ranging from sarcopenia and cognitive impairment to heart failure and metabolic disorders. Early experimental work indicates that cells receiving fresh mitochondria show restored ATP production, reduced oxidative stress markers, and improved functional capacity. This approach offers particular promise for tissues with high metabolic demands that show pronounced age-related decline, including skeletal muscle, cardiac tissue, and neural networks. Unlike systemic pharmaceutical interventions that attempt to support existing damaged mitochondria, direct replacement strategies aim to fundamentally reset cellular energy metabolism. The technology also enables personalized treatment protocols, as devices can be calibrated to deliver specific mitochondrial doses based on tissue type, patient age, and degree of cellular dysfunction. Industry observers note that this represents a shift from managing symptoms of aging to addressing one of its root biological mechanisms.
Current development efforts focus on refining device biocompatibility, optimizing mitochondrial viability during processing and delivery, and establishing protocols for safe clinical translation. Prototype systems are being tested in laboratory settings to validate mitochondrial uptake efficiency and measure functional improvements in aged tissue models. The technology faces several technical hurdles, including maintaining organelle integrity throughout the harvesting and delivery process, ensuring adequate cellular uptake in target tissues, and preventing immune responses to transferred mitochondria. Researchers are exploring various delivery modalities, from single-use catheter-based systems for cardiac applications to chronically implanted devices that could provide periodic mitochondrial supplementation to skeletal muscle. As the field of cellular rejuvenation advances, mitochondrial augmentation devices may emerge as a cornerstone technology for treating age-related tissue degeneration, potentially extending healthspan by restoring the fundamental energy-generating capacity that declines with age. The convergence of microfluidics, organelle biology, and minimally invasive delivery systems positions this approach at the frontier of regenerative medicine's effort to reverse rather than merely slow the aging process.
Related Organizations
Develops Mitochondrial Augmentation Therapy (MAT), involving the enrichment and manufacturing of autologous cells.
Developing Therapeutic Mitochondria Transplantation (TMT) for ischemia-reperfusion injury and aging.
Startup developing 'mitlets' (mitochondrial transfusion) to replenish mitochondria in aging bodies.
Conducts advanced research on cross-circulation platforms to rehabilitate donor lungs and livers ex vivo.
Developed the SWIFT (Sacrificial Writing into Functional Tissue) method for 3D printing vascular channels in living matrices.
Clinical-stage biotech developing therapies to treat mitochondrial dysfunction.
Major pharmaceutical company that acquired Iota Biosciences.
Focuses on restoring mitochondrial function to reverse aging, specifically starting with hair and skin.
Leader in exosome technology for delivery of therapeutics.