Bioelectric Morphogenetic Arrays
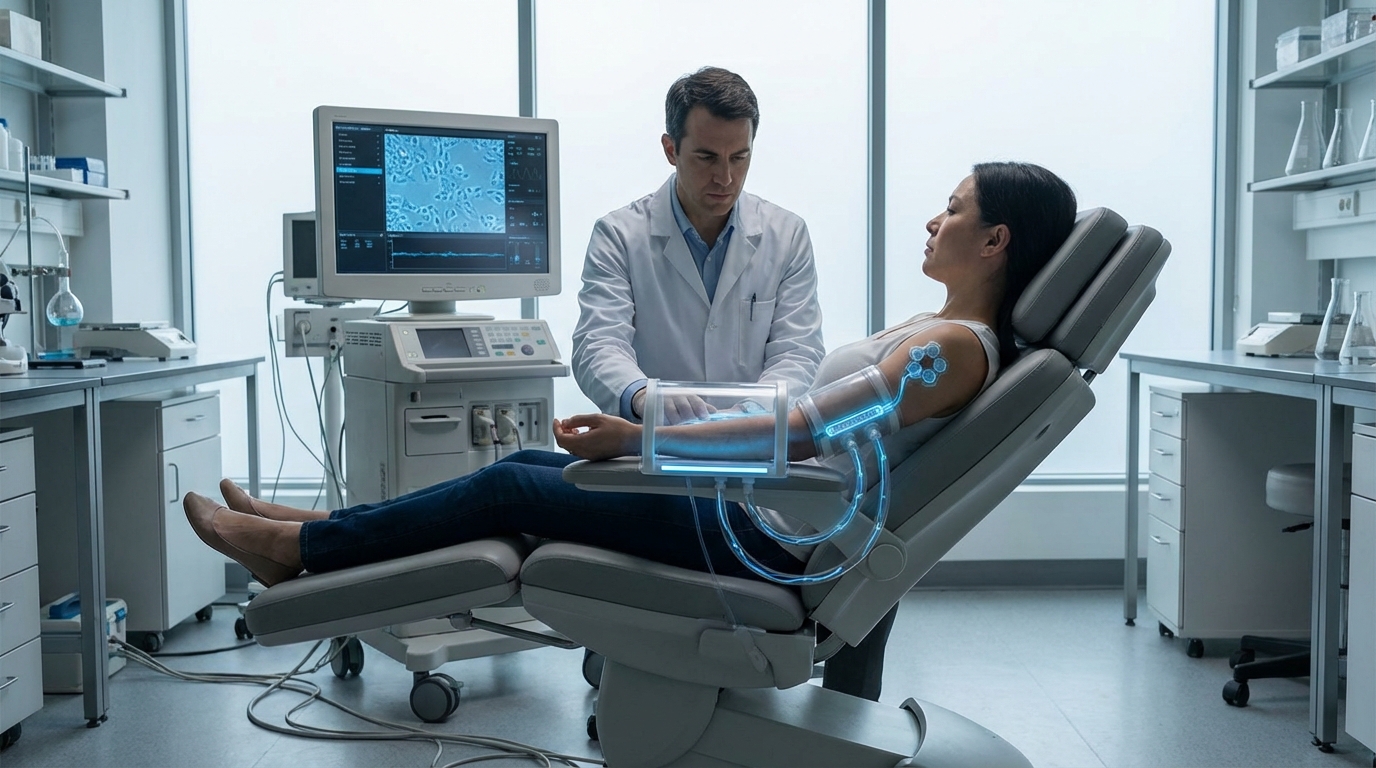
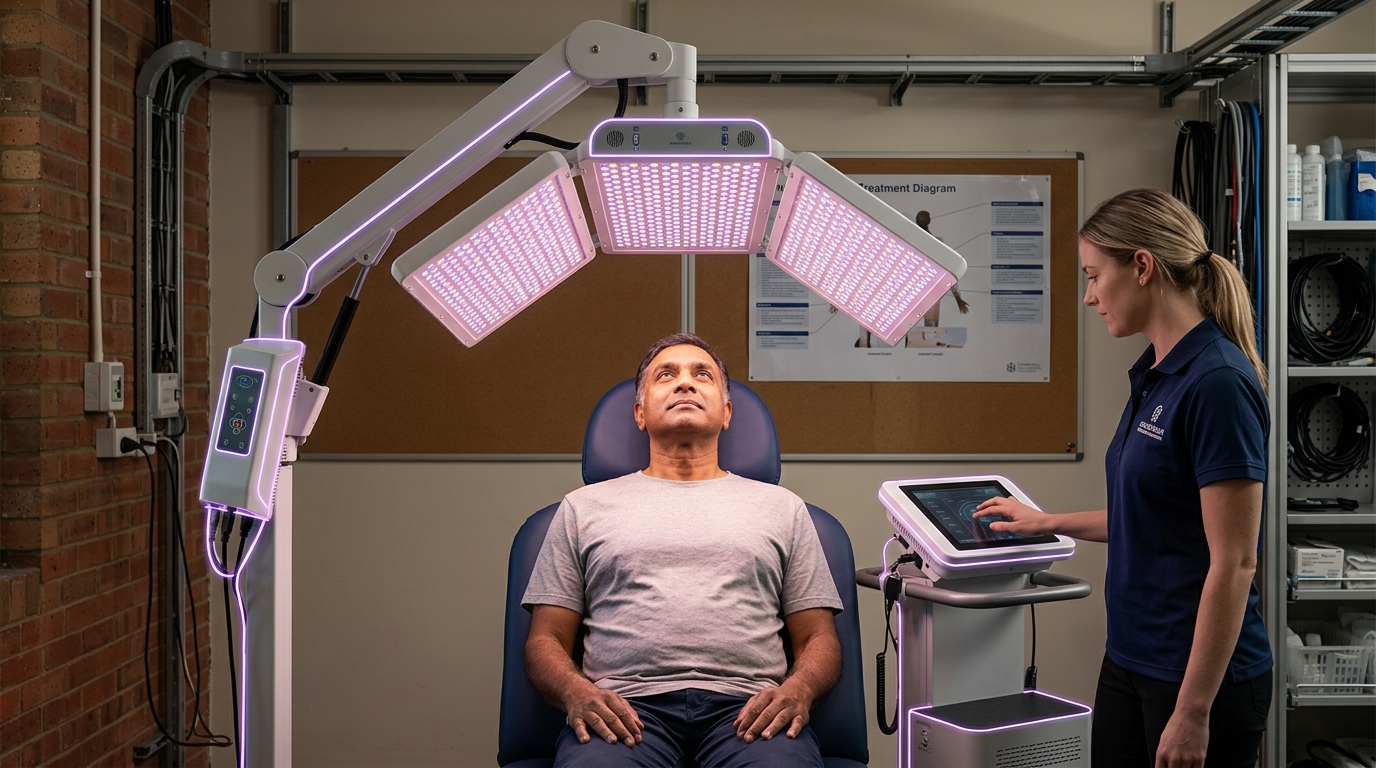
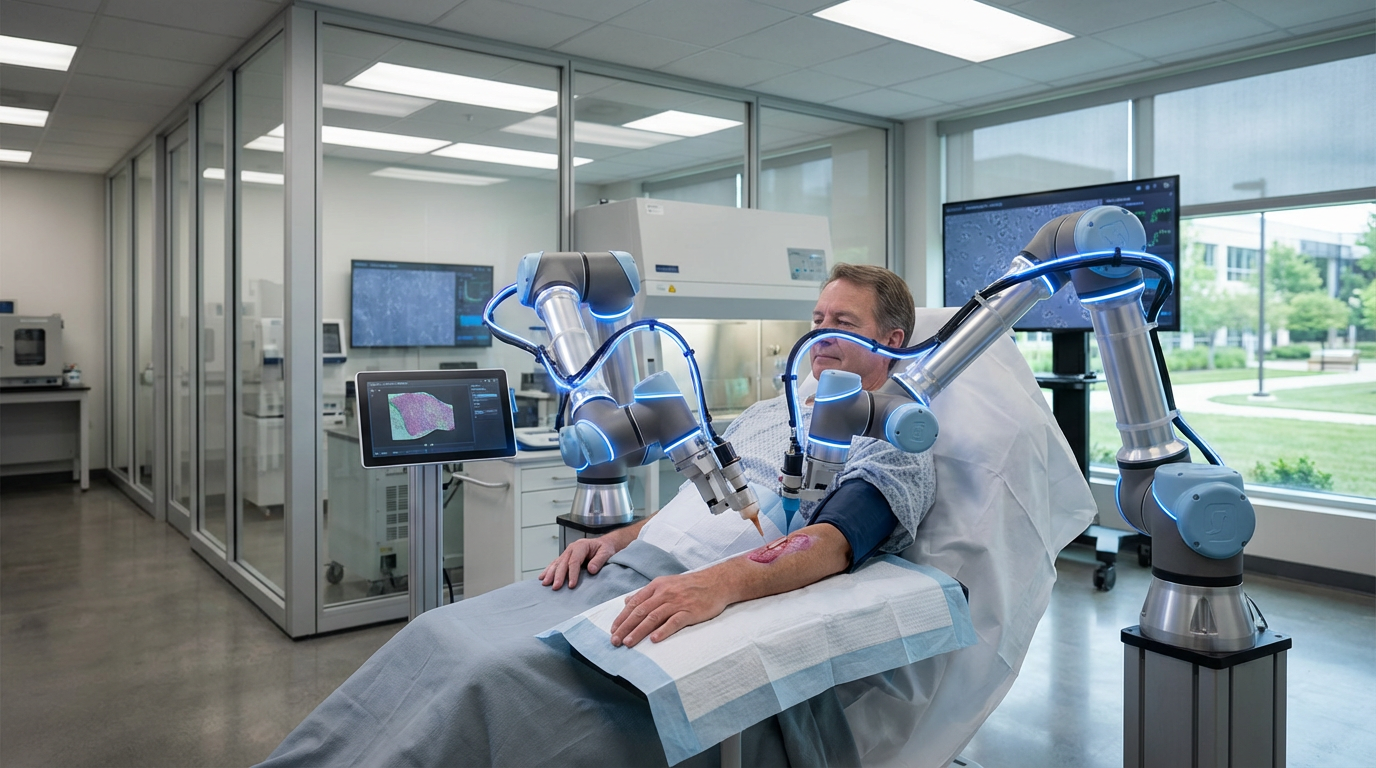
Bioelectric morphogenetic arrays represent a fundamental shift in regenerative medicine, moving beyond traditional biochemical approaches to harness the electrical signals that govern cellular behavior and tissue architecture. These sophisticated devices—ranging from precision electrode grids to light-activated optogenetic interfaces—detect and manipulate the voltage gradients that exist across cell membranes and tissue boundaries. Research has revealed that cells communicate not only through chemical messengers but also through bioelectric patterns, with specific voltage states acting as instructional codes that determine whether a group of cells should form an eye, a limb, or remain undifferentiated. The arrays work by measuring the resting membrane potential across wound sites or developing tissues, then applying targeted electrical stimulation or light pulses to shift cells into voltage states associated with regeneration rather than scarring. This approach draws on observations from naturally regenerative organisms like salamanders and planarian worms, where bioelectric signals orchestrate the rebuilding of entire body structures with remarkable fidelity.
The promise of bioelectric morphogenetic control addresses a critical limitation in current regenerative therapies: the inability to guide stem cells or tissue scaffolds into forming complex, properly organized structures. While stem cell treatments can provide raw cellular material, they often lack the spatial information needed to recreate the intricate architecture of organs or appendages. Bioelectric arrays offer a solution by providing the morphological blueprint that cells need to self-organize into functional tissues. Early experimental work suggests that manipulating bioelectric patterns can trigger regenerative programs in mammals, which typically lack the robust regenerative capabilities of simpler organisms. This technology could potentially overcome the scarring response that normally prevents human tissue from regenerating after injury, instead activating developmental pathways that guide cells to rebuild lost or damaged structures. The approach also offers advantages over purely genetic interventions, as bioelectric signals can be applied temporarily and adjusted in real-time, reducing the risks associated with permanent genetic modifications.
Current research efforts are exploring applications ranging from limb regeneration in amputation cases to the restoration of damaged organs and the treatment of birth defects. Laboratory demonstrations have shown that bioelectric manipulation can induce the formation of additional limbs in model organisms and influence the development of complex structures like eyes in unexpected locations, proving that voltage patterns carry powerful morphogenetic information. As the field advances, researchers are working to decode the specific bioelectric signatures associated with different tissue types and developmental stages, creating libraries of voltage patterns that could be replicated to guide human regeneration. The integration of bioelectric arrays with other regenerative technologies—including biomaterial scaffolds, growth factors, and cellular therapies—points toward combination approaches that leverage multiple pathways to tissue reconstruction. This convergence aligns with broader trends in regenerative medicine toward precision control of biological processes, suggesting that future treatments may routinely employ bioelectric programming alongside traditional pharmaceutical and surgical interventions to restore form and function to damaged tissues.
Related Organizations
Led by Michael Levin, this lab is the world leader in cracking the bioelectric code of morphogenesis.
A spin-off from Tufts University developing bioelectric interventions for limb regeneration and organ health.
Developed the SWIFT (Sacrificial Writing into Functional Tissue) method for 3D printing vascular channels in living matrices.
Partnership between GSK and Verily to develop bioelectronic medicines.
Commercializing 'neural dust' technology: millimeter-sized ultrasonic implants.
Developers of the CellFX System, which uses Nano-Pulse Stimulation (NPS) technology to clear cellular lesions with high precision.
Bioelectronic medicine company transforming the standard of care for chronic disease.
Developing a platform to treat chronic inflammation via the vagus nerve.
Commercializes bioelectronic devices for non-invasive treatment of sinus and inflammatory conditions.