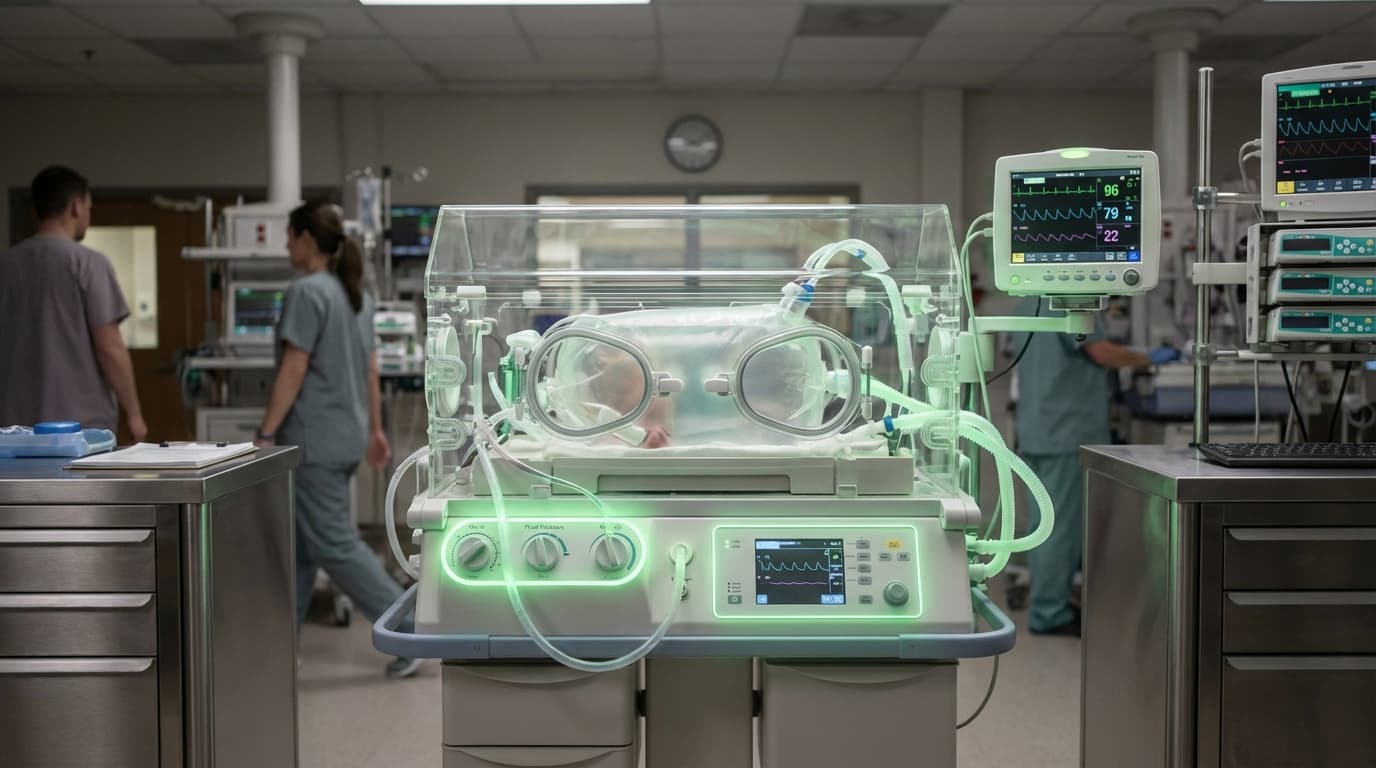
Artificial Wombs
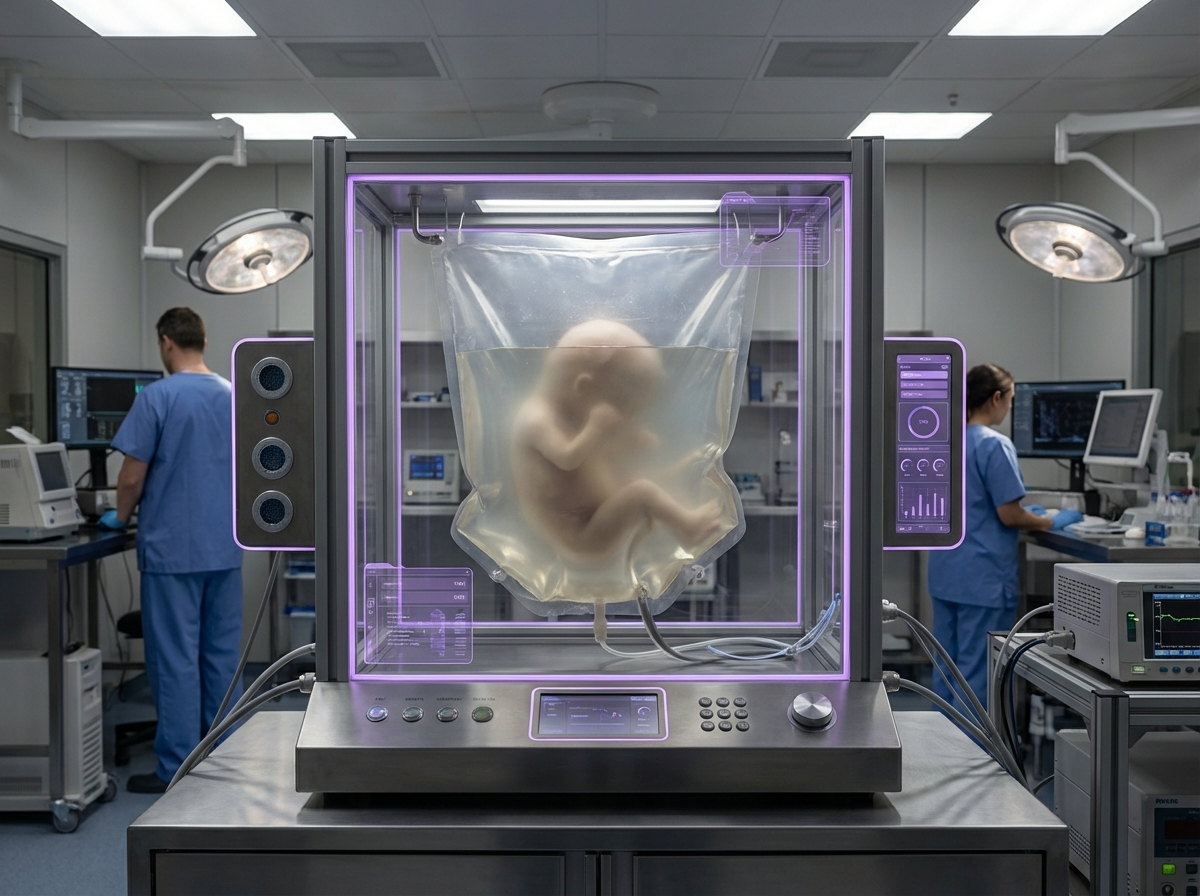
Artificial womb technology represents a breakthrough in neonatal care, designed to provide life-support for extremely premature infants by replicating the intrauterine environment outside the mother's body. The core system, often referred to as a biobag, consists of a sterile, temperature-controlled enclosure filled with synthetic amniotic fluid that surrounds the developing infant. An external oxygenator connects to the infant's umbilical cord, performing the gas exchange function normally handled by the placenta, while carefully regulated fluid circulation maintains optimal temperature, nutrient delivery, and waste removal. The closed system protects the fragile infant from infection and external stimuli while allowing continuous monitoring of vital signs and development. Unlike traditional incubators that rely on the infant's immature lungs to breathe air, artificial wombs enable the continuation of fluid-based respiration, allowing critical organ systems—particularly the lungs and brain—to develop more naturally before the infant must adapt to breathing air.
The technology addresses one of neonatal medicine's most pressing challenges: the survival and healthy development of infants born at the threshold of viability, typically between 22 and 24 weeks of gestation. At these early stages, underdeveloped lungs, fragile blood vessels, and immature organ systems make survival extremely difficult, and those who do survive often face lifelong complications including chronic lung disease, cerebral palsy, and developmental delays. Current neonatal intensive care relies on mechanical ventilation, which can damage delicate lung tissue, and incubators that expose premature skin and organs to air before they are ready. Artificial wombs offer the potential to bridge the critical weeks of development in an environment that more closely mimics natural gestation, potentially reducing the severe complications associated with extreme prematurity. This capability could transform outcomes for the thousands of families affected by premature birth each year, while also reducing the substantial long-term healthcare costs associated with prematurity-related complications.
Research institutions have successfully demonstrated artificial womb technology in animal models, with lamb fetuses developing normally for several weeks within biobag systems. These proof-of-concept studies have shown promising results in lung maturation and overall development, suggesting the technology's potential for human application. However, the transition to human trials faces significant regulatory, ethical, and technical hurdles, including questions about the appropriate gestational age for intervention, the duration of support, and long-term developmental outcomes. Early clinical applications, when approved, are likely to focus on the most critically premature infants who currently have minimal survival chances with conventional care. The technology aligns with broader trends in regenerative medicine and bioengineering, where the boundaries between natural biological processes and technological support continue to blur. As research progresses and safety data accumulates, artificial wombs may eventually extend the lower limit of viability, offering hope to families facing extremely premature births while raising important societal questions about the nature of gestation, parental bonding, and the beginning of life outside the womb.
Related Organizations
Developed the 'Biobag' system (EXTEND), demonstrating successful support of premature lamb fetuses in an artificial womb environment.
A spin-out company working to commercialize the EXTEND artificial womb technology developed at CHOP for extreme premature infants.
EVE Therapy
Australia · Startup
Commercializing the EVE (Ex-vivo Uterine Environment) therapy platform developed at the University of Western Australia.
The regulatory body convening advisory committees to discuss the safety, efficacy, and ethics of artificial womb technology (EXTEND).
Researchers here developed the EVE therapy platform, an ex-vivo uterine environment for treating preterm infants.
Leads the Perinatal Life Support (PLS) consortium, developing an artificial womb system using simulation mannequins.
An independent body that examines ethical issues in biology and medicine, actively publishing on the ethics of artificial wombs.
Research into spintronics and probabilistic computing which overlaps with reversible logic principles.