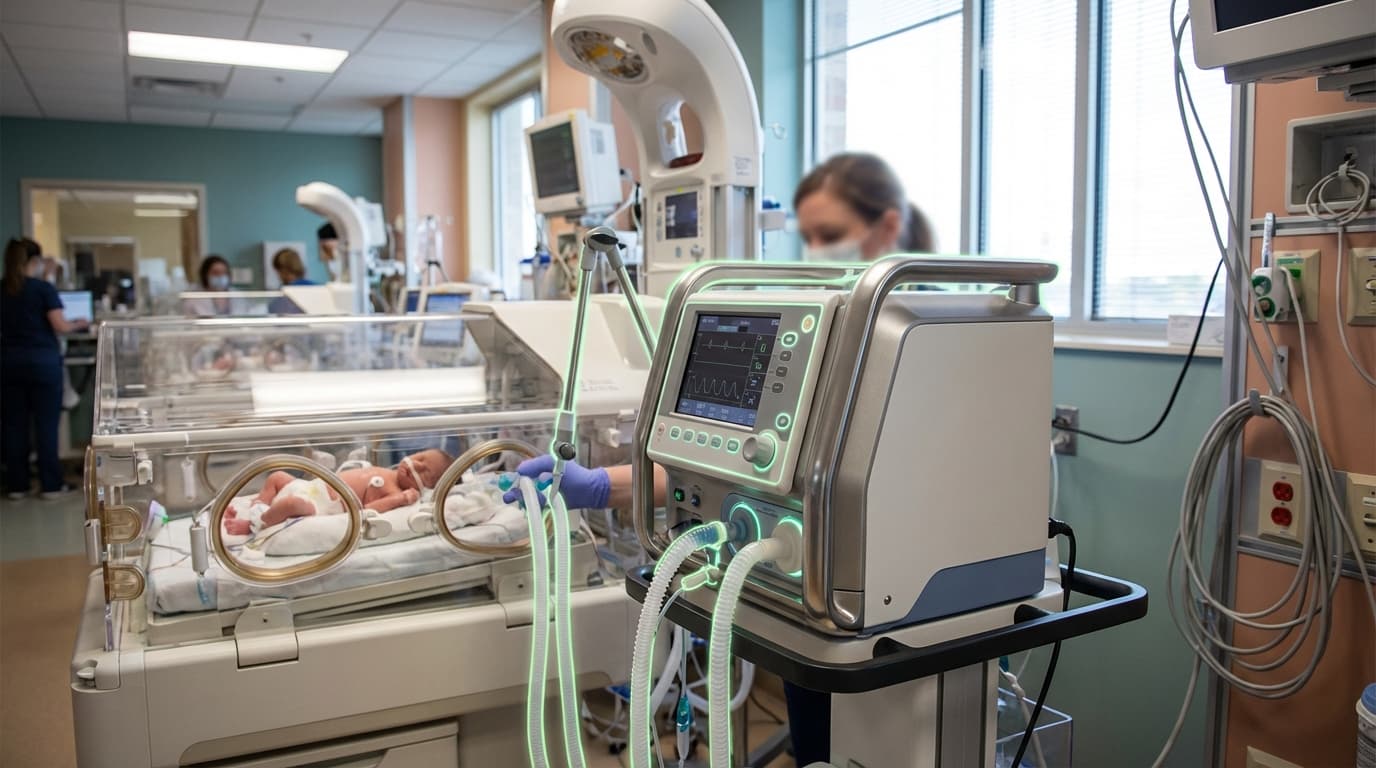
Closed-Loop Neonatal Ventilation
Closed-loop neonatal ventilation represents a significant advancement in respiratory support for preterm and critically ill newborns, whose underdeveloped lungs are extremely vulnerable to both insufficient oxygen delivery and ventilator-induced injury. Traditional mechanical ventilation in neonatal intensive care units (NICUs) requires constant manual adjustment by clinicians, who must balance the competing risks of hypoxia, hyperoxia, and barotrauma—all of which can lead to serious complications including bronchopulmonary dysplasia, retinopathy of prematurity, and neurodevelopmental impairment. These closed-loop systems employ sophisticated algorithms that continuously monitor multiple physiological parameters, including tidal volume, respiratory rate, oxygen saturation, and transcutaneous carbon dioxide levels, then automatically adjust ventilator settings such as peak inspiratory pressure, positive end-expiratory pressure, and inspired oxygen concentration in real time. The technology essentially creates a feedback loop where the ventilator responds dynamically to the infant's changing respiratory needs, much as a healthy lung would self-regulate, but with the precision and consistency that human monitoring cannot sustain around the clock.
The neonatal care industry faces a critical challenge in managing the delicate respiratory needs of extremely preterm infants, particularly those born before 28 weeks of gestation whose lungs may be profoundly immature. Manual ventilation management is labor-intensive, requires highly specialized expertise, and is subject to delays in response time and variability between different clinicians and shifts. Research suggests that even brief periods of suboptimal ventilation can have lasting consequences for these vulnerable patients. Closed-loop systems address these limitations by providing consistent, protocol-driven care that responds within seconds to physiological changes, potentially reducing the cognitive burden on NICU staff while simultaneously improving patient outcomes. Early clinical studies indicate that automated ventilation may reduce the duration of mechanical ventilation, decrease the incidence of severe complications, and improve the consistency of care across different times of day and staffing levels. This technology also enables more precise titration of oxygen therapy, which is particularly crucial given that both oxygen deprivation and excess oxygen exposure carry serious risks for developing organs.
Several closed-loop neonatal ventilation systems have progressed from research prototypes to clinical evaluation in leading NICUs, with some receiving regulatory clearance in various markets. These systems are being deployed in tertiary care centers with high-volume neonatal units, where they complement existing monitoring infrastructure and integrate with electronic health records to provide comprehensive respiratory management. The technology aligns with broader trends in neonatal care toward precision medicine and individualized treatment protocols, recognizing that each preterm infant presents unique physiological characteristics that require tailored support. As artificial intelligence and machine learning capabilities continue to advance, future iterations of closed-loop ventilation may incorporate predictive analytics to anticipate respiratory decompensation before it occurs, or adapt their algorithms based on individual patient response patterns over time. The growing emphasis on reducing healthcare-associated complications and optimizing long-term neurodevelopmental outcomes for preterm infants positions automated respiratory support as an increasingly essential component of modern neonatal intensive care, particularly as survival rates for extremely preterm infants continue to improve and attention shifts toward minimizing morbidity among survivors.
Related Organizations
A specialist in neonatal ventilation that developed 'OxyGenie', a closed-loop control system for automatic regulation of inspired oxygen (FiO2) in neonates.
Global medical technology company known for the Servo ventilator line, featuring Neurally Adjusted Ventilatory Assist (NAVA).
Manufacturer of intelligent ventilation solutions, including INTELLiVENT-ASV, which automatically adjusts ventilation and oxygenation.
International leader in the fields of medical and safety technology.
Manufacturer of the Leoni Plus neonatal ventilator, which includes CLAC (Closed-Loop Automatic Oxygen Control).

Murdoch Children's Research Institute
Australia · Research Lab
Largest child health research institute in Australia, conducting clinical trials on automated oxygen control in preterm infants.
Develops ultrasound systems (Voluson) with AI features that assist in assessing fetal development and cervical length, key indicators for preterm risk.
Global medical device developer offering the SV series ventilators with adaptive ventilation modes.
Japanese medical electronics company producing the NKV-550 ventilator series with protective lung applications.













