Indigenous Surgical Robot Systems
Geography: Asia Pacific · East Asia · South Korea
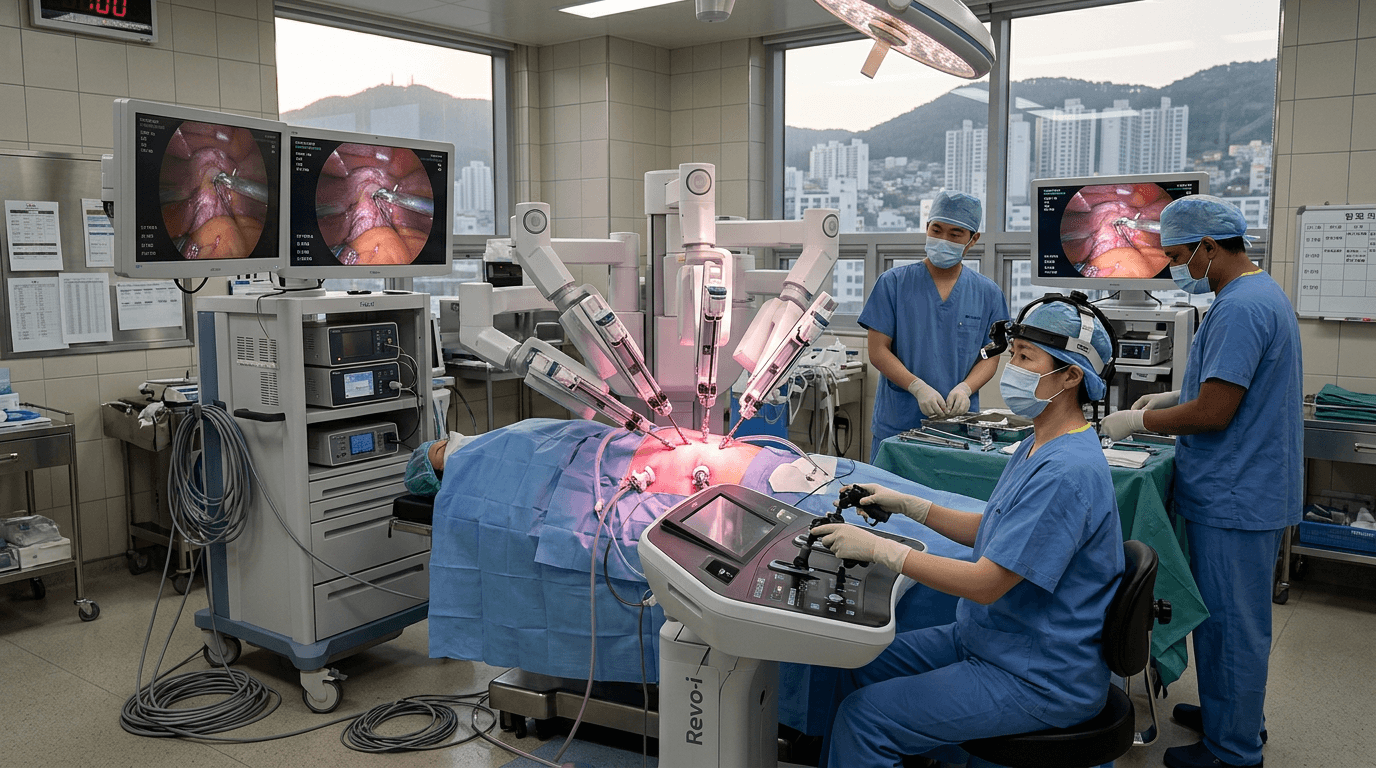
Meerecompany's Revo-i robotic surgical system received Korean MFDS approval in 2018 as the world's second regulatory-approved laparoscopic surgical robot, directly competing with Intuitive Surgical's da Vinci system that has monopolized the $7B+ surgical robotics market for two decades. Clinical trials including pancreatic surgery demonstrated Revo-i's safety and efficacy are comparable to da Vinci, with the critical difference being significantly lower acquisition and per-procedure costs — making robotic surgery accessible to hospitals that cannot afford the $1-2 million da Vinci system.
The Revo-i system features four robotic arms, a 3D HD endoscopic camera, and 7-degrees-of-freedom instruments with wristed articulation. The surgeon operates from a console with motion scaling and tremor filtering. Clinical studies published in peer-reviewed journals confirmed that Revo-i performed comparably to da Vinci in procedures including prostatectomy, hysterectomy, and pancreaticoduodenectomy — some of the most technically demanding minimally invasive surgeries.
Korea's entry into surgical robotics follows the same market-disruption pattern seen in biosimilars: identify a high-margin market dominated by a single player, reverse-engineer comparable technology, and offer it at a fraction of the price. Revo-i is expanding internationally, targeting markets in Asia, the Middle East, and Eastern Europe where da Vinci's pricing makes robotic surgery uneconomical. If Meerecompany scales production and gains regulatory approvals in the EU and US, it could do to surgical robotics what Celltrion did to biologic drugs — force dramatic price reductions through competition.