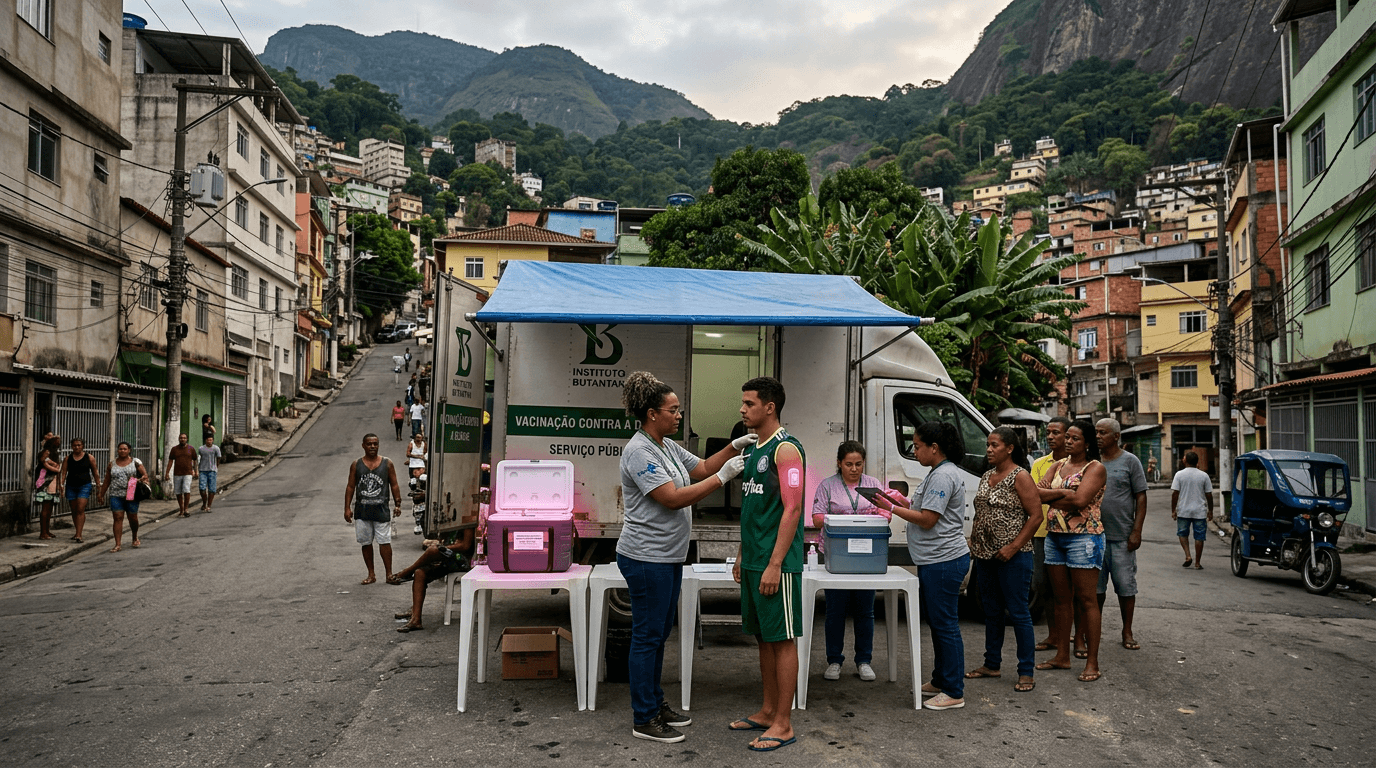
Single-Dose Dengue Vaccine (Butantan-DV)
Geography: Americas · South America · Brazil
Butantan-DV is a tetravalent live-attenuated dengue vaccine requiring only a single dose — a critical advantage over Sanofi's Dengvaxia (3 doses) and Takeda's Qdenga (2 doses). ANVISA granted approval in November 2025 after a Phase III trial enrolling 16,000+ participants aged 2-59 across Brazil. Five-year follow-up data published in Nature Medicine (March 2026) showed 80.5% efficacy against severe dengue with warning signs and 74.7% overall efficacy.
Dengue infects an estimated 390 million people annually worldwide, with Brazil recording nearly a million cases in early 2024 alone. A single-dose vaccine is transformative for public health logistics: it eliminates the dropout problem of multi-dose regimens, critical for reaching remote Amazon communities where follow-up visits are impractical. Unlike Dengvaxia, Butantan-DV is effective regardless of prior dengue exposure (serostatus), removing the need for pre-vaccination screening.
Brazil developed this vaccine domestically through Butantan Institute's 20+ year research program, with technology licensed from NIH but adapted and clinically developed entirely in Brazil. Production will scale through Butantan's existing manufacturing base — already one of the largest vaccine producers in the Southern Hemisphere. Gavi has identified it as a potential game-changer for dengue control across the tropics, serving 4 billion people in endemic regions.