Dynamic Consent Orchestration
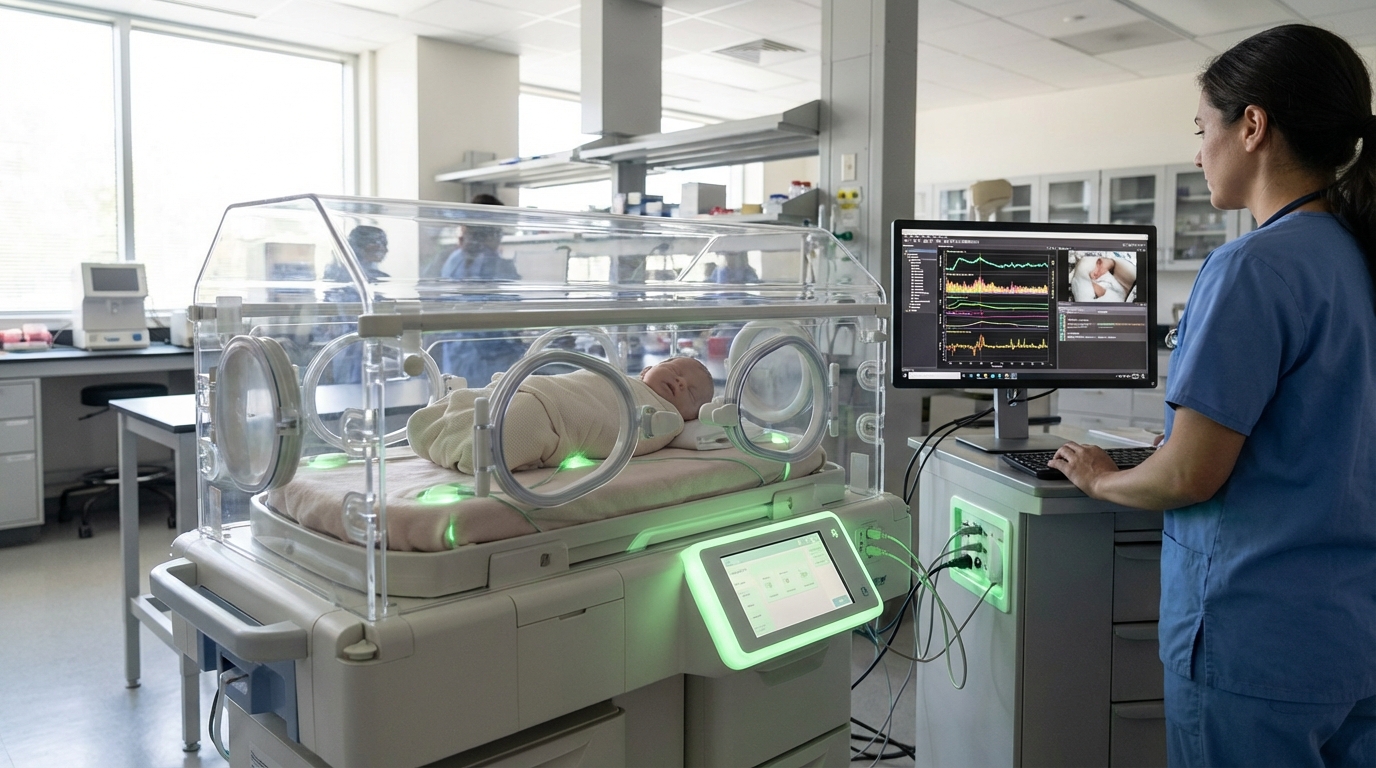
Dynamic Consent Orchestration represents a paradigm shift in how families manage sensitive health and developmental data throughout the reproductive journey and early childhood. Unlike traditional static consent forms that grant blanket permissions at a single point in time, this technology enables continuous, granular control over who can access, use, and share information ranging from fertility treatment records and genetic screening results to neonatal health data and developmental milestones. The system operates through secure digital platforms that maintain detailed audit trails of all data access and usage, allowing parents to modify permissions in real-time as circumstances change. At its technical core, these platforms employ sophisticated identity management systems, blockchain-based consent ledgers, and role-based access controls that can distinguish between different types of data recipients—from healthcare providers and researchers to insurance companies and educational institutions. The architecture typically includes user-friendly interfaces that translate complex privacy options into understandable choices, while maintaining compliance with evolving data protection regulations across different jurisdictions.
The fertility and perinatal care industries face mounting challenges around data governance, particularly as genetic testing, assisted reproduction, and precision medicine generate increasingly detailed information about families before children are even born. Traditional consent models create significant problems: parents may unknowingly grant permanent access to sensitive information during vulnerable moments, children have no say in decisions made about their data before they can consent, and families lack mechanisms to respond to new research findings or changing family circumstances. Dynamic Consent Orchestration addresses these limitations by introducing temporal controls that can evolve as children mature, allowing for age-appropriate transitions of consent authority from parents to young adults. This capability is particularly crucial in contexts like biobanking, where samples and data collected at birth might be used for research purposes decades later. The technology also enables families to selectively share information—for instance, permitting anonymised data for research while restricting identifiable information, or allowing access for specific medical purposes while blocking commercial uses.
Early implementations of dynamic consent systems have emerged primarily in research contexts, where institutions conducting longitudinal studies of child development and family health have piloted platforms that allow ongoing participant engagement with their data. These deployments indicate that families value the ability to revisit consent decisions, particularly as children reach developmental milestones or when new types of data usage become possible through technological advances. The approach aligns with broader trends toward patient-centered care and data sovereignty, reflecting growing recognition that individuals and families should maintain meaningful control over their health information throughout the lifespan. As precision medicine and personalised pediatric care become more prevalent, the capacity to manage consent dynamically will likely transition from an ethical best practice to a regulatory requirement, fundamentally reshaping how the healthcare industry approaches data governance for vulnerable populations and establishing new standards for respecting family autonomy across generations.